Internal and External Validity
Source: 1
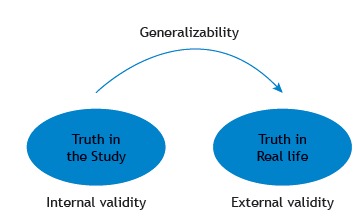
Internal Validity
Internal validity is defined as the extent to which the observed results represent the truth in the population we are studying, meaning that we do not introduce methodological errors. The internal validity can be threatened by many factors, such as errors in measurement or in the selection of participants in the study.
- High internal validity means less confounding variables exist in the experiment
- High internal validity means the results of experiments are caused only by independent variables
- High internal validity is achieved via using random sampling and control variables
- Control variables are variables that can impact the dependent variables and thus are kept constant in the experiment.
Non-response Bias
Therefore our results could suffer from a potential ‘non-response bias’ (i.e., the opinions of the respondents who chose to participate may be different from who did not)
Source to read more: J Scott Armstrong and Terry S Overton. 1977. Estimating nonresponse bias in mail surveys. Journal ofMarketing Research 4, 3 (1977), 396–402
External Validity
External validity of the study refers to the extent to which the results of a study are generalizable to patients in our daily practice, especially for the population that the sample is thought to represent.
Trouble with the lack of validity
The lack of internal validity implies that the results of the study deviate from the truth, and therefore, we cannot draw any conclusions. Hence, if the results of a trial are not internally valid, external validity is irrelevant.
The lack of external validity implies that the results of the trial may not apply to patients who differ from the study population and, consequently, could lead to low adoption of the treatment tested in the trial by other clinicians.